Air Pollution XII
Editor
C.A.Brebbia
Wessex Institute of Technology, UK
S. A. Grinshpun, A. Adhikari, B. U. Lee, M. Trunov, G. Mainelis, M. Yermakov & T. Reponen
Center for Health-Related Aerosol Studies, Department of Environmental Health, University of Cincinnati, USA
Current affiliation: Mechanical Engineering Department, New Jersey Institute of Technology, USA
Current affiliation: Department of Environmental Sciences, Rutgers University, USA
Abstract
Various health effects are associated with or directly caused by respirable airborne particles and microbial agents. To reduce the hunan exposure to these indoor pollutants, numerous techniques have been developed over the years. In this study, we have investigated the effect of unipolar air ionization on airborne dust particles and microorganisms in indoor environments. The concentration and particle size distribution were measured in real time using optical and aerodynamic particle counters with a special focus on the bacterial particle size range of 0.5 to 2 µm. The tests were conducted in three indoor chambers of different volumes (ranging from 26 L to 24.3 m3) at different ion emission rates (producing air ions at ~104 to ~l05 ions/cm3 as measured at ~1 m from the source). The concentration decay occurring due to ionic emission was compared to the natural decay for four types of challenge aerosols. Resulting from the interaction with unipolar air ions, airborne particles exhibited considerable electric charges of the same polarity as the emitted ions. Due to electrostatic repelling forces, the particles migrated toward the indoor surfaces and rapidly deposited on these surfaces. Two small, battery operated ionic emitters tested in this study showed significant air cleaning efficiency for respirable (sub- and super-micrometer) particles. This effect was more pronounced in smaller air volumes. The efficiency of ion emission in reducing the viability of airborne microorganisms in indoor air was also evaluated in a specially designed set-up. Two species of Gram-negative bacteria (Pseudomonas fluorescens and Escherichia coli) and one species of Gram-positive bacteria ( Staphylococcus epidermidis) were tested. It was found that a significant percentage of airborne viable bacteria could be inactivated by the ion emission: up to 92% of E. coli was inactivated during a one-minute exposure in dry air. It was concluded that the ion-driven decrease in the aerosol concentration combined with the bactericidal effect can significantly reduce human exposure to indoor air pollutants, such as particles and microorganisms.
Introduction
Respirable airborne particles, including airborne dust, microbial agents and aeroallergens, may cause adverse health effects, such as asthma, allergic diseases (2, 7, 10, 14] and airborne infections [2]. Although health effects associated with biological aerosols have been of a special concern for decades, recent outbreaks of emerging infections as well as the growing concern about bioterrorism have drawn additional attention to the development of control methods against indoor air pollutants, particularly against viable bacterial cells and spores.
The deposition and retention of particles larger than 0.5 µmin upper and lower parts of the respiratory tract (and subsequently the health effects associated with these particles) depends on the aero.dynamic particle size (dae). For most of the airborne bacterial species, dae ranges from 0.5 to 2 µm. For example, our earlier measurements of aerosolized bacteria using aerodynamic particle counters have shown dae=0.7-0.8 µm for Pseudomonas fluorescens [15, 17], 0.9 µm for Bacillus subtilis [17], and 1.10 µm for Micrococcus luteus [15]. The optical particle size of bacterial particles is usually close to the aerodynamic one (within ±20%) [l]. Bacteria aerosolized from liquids (e.g., saliva or mucus) may initially be carried in or by larger droplets. However, the water content is likely to evaporate rapidly, thus decreasing the particle size to 1-2 µm. This is almost always true in indoor air environments (as long as the atmosphere is not close to the saturation level).
Among several air cleaning techniques currently used for reducing the aerosol concentration in indoor air settings (e.g., residential and office rooms, as well as aircraft and automobile cabins), the air ion emission has been explored and shown a promise [3, 4, 6]. The data obtained in two latest studies conducted by our research team [5, 8] helped to better understand the mechanisms involved in indoor air purification due to unipolar ion emission. The principle is schematically shown in Figure 1. Resulting from their interaction with unipolar air ions, the airborne particles carry considerable positive or negative electric charges, depending on the polarity of the emitter. Due to electrostatic repelling forces, the particles migrate toward the indoor surfaces and rapidly deposit on these surfaces.
The air ionization has been incorporated into commercial air purification devices manufactured by Ionair, Inc. (Midland, Ml, USA), Sharp Corporation (Osaka, Japan), Topway Electronic Factory Company (Guangzhou, China), Wein Products, Inc. (Los Angeles, CA, USA) and other companies. Most of the ionic air purifiers, including unipolar ion emitters, have been originally designed to reduce the exposure to all types of aerosol particles, irrespective of their biological properties. Since viable airborne bacteria represent a specific hazard, it is important to determine the physical and biological efficiencies of ionic air purifiers against these air contaminants. Toe physical efficiency of ionic air purifiers represents their ability to reduce the concentration of bacterial particles in the air, whereas the biological (bactericidal) efficiency represents their ability to reduce the microbial viability by inactivating viable microorganisms that remain airborne. The overall air cleaning efficiency is a product of the physical and bactericidal efficiencies of the ionic air purifier. In this study, we evaluated both the physical and bactericidal effects of unipolar ion emission against viable bacterial cells.
Experimental design and method
Physical efficiency
The air cleaning process was experimentally investigated by measuring the aerosol concentration and particle size distribution in real time inside indoor test chambers of three different air volumes. These included a large walk-in chamber that simulated a residential room (24.3 m3), a small walk-in chamber that simulated a small office area or an automobile cabin (2.6 m3), and a "box" that simulated a small enclosure (26 L). Polydisperse NaCl aerosol monodisperse PSL spheres of three sizes, polydisperse smoke aerosol, and vegetative cells of Pseudomonas fluorescens bacteria (ATCC 13525) were used as the challenge aerosols in the experiments. The sodium chloride and PSL particles, as well as bacterial cells were generated by 3-jet Collison nebulizer (BGI, Inc., Waltham, MA, USA) operated at a pressure of 12 psi. Smoke particles were generated by a custom-built smoke generator. The challenge aerosol was delivered into the test chamber with a clean, laboratory-filtered air at a specific temperature (T = 22°C) and relative humidity (RH= 28±5%).

Two types of instruments were used for the particle size selective measurements. The Grimm optical particle counter (OPC, model 1.108, Grimm Technologies Inc., Douglasville, GA, USA) served as an optical size spectrometer in all three test chambers. The Aerosizer (API/TSI, Inc. St. Paul, MN, USA) and the Electrical Low Pressure Impactor (ELPI, TSI Inc./Dekati Ltd, Tampere, Finland) served as aerodynamic particle sizers (Aerosizer operated in parallel to the Grimm OPC in the small walk-in chamber, whereas the ELPI was used in the large walk-in chamber). Although the operational particle size ranges of the Grimm OPC, Aerosizer and ELPI are distinctly different, all three are capable of accurately measuring the particle concentration within the size range of bacterial particles, i.e., 0.5 - 2 µm.
The experimental facility used to study the physical efficiency of ion emitters is schematically shown in Fig. 2. Table 1 describes the characteristics of the test chambers and lists the challenge aerosols and the instrumentation involved in the tests.
The concentration decay occurring due to ionic emission was compared to the natural decay. The nondimensional particle concentration was determined as a ratio of the concentration measured at a specific time point, t, to the initial one (measured at t = 0). The tests were performed under calm air conditions.
In this paper, we report the data obtained with two portable (wearable) ion emitters available from Wein Products, Inc.: Minimate, AS180i and AS150MM*. The y produce positive air ions at essentially different ion emission rates. To quantify these rates in terms of the volumetric ion density, the air ion concentration created inside the 24.3 m3 chamber was determined with an Air Ion Counter (AlpbaLab Inc., Salt Lake City, UT, USA) at specific distances from the ion source.
Bactericidal efficiency
Figure 3 schematically . shows the experimental facility developed for investigating the effect of air ions emitted by an ionic air purifier on the viability of airborne bacteria. The setup consisted of the following major elements: autoclavable bioaerosol test chamber (LxWxH = 60x30x30 cm3) made of metal, source of HEPA filtered air, air temperature and humidity control system, Collison nebulizer, Grimm OPC, and BioSampler (SKC Inc., Eighty Four, PA, USA). The dimensions of the chamber represent the breathing zone. The Collison nebulizer, commonly used for aerosolizing bacteria from liquid suspensions, simulated the aerosolization of viable bacteria by human coughing and sneezing. The OPC monitored the particles throughout the chamber air volume to assure a uniform aerosol concentration pattern. The BioSampler provided accurate and representative collection of viable bacteria from the air into liquid for subsequent total counting and colony forming unit (CFU) enumeration [18). In these experiments, the bacteria were airborne for approximately one minute between the microbial aerosolization and the sampling. Based on our preliminary studies, this time was sufficiently short to minimize the effect of desiccating air on bacterial viability.
The entire setup was housed in a Class II, Type B2, biological safety cabinet (Sterilchem GARD, Baker Company, Sanford, ME, USA). This allowed us to maintain sterile conditions during the experiments and to ensure that all airborne bacteria were properly exhausted after passing through the bioaerosol test chamber. The ionic air purifier under the test was placed at the chamber entry point, downstream of the nebulizer outlet. The ions produced by the air purifier were carried by the temperature- and humidity-controlled sheath air flow (36 L/min) that entered the chamber at the same point. The OPC, the BioSampler, and the humidity/temperature meter (portable thermohygrometer pen, Fisher Scientific, Pittsburgh, PA, USA) were placed downstream, close to the outlet of the chamber. The walls of the chamber and all the equipment units were grounded to avoid any electric charge build-up inside the experimental setup.
Similar to the evaluation of the physical efficiency, two models of Wein wearable ionic air purifiers, Minimate, AS180i and AS150MM*, were tested with respect to the bactericidal efficiency.
Two species of Gram-negative bacteria (Pseudomonas fluorescens and Escherichia coli) and one species of Gram-positive bacteria (Staphylococcus epidermidis) were utilized in this part of the study as challenge aerosols. Cultures of P. fluorescens (ATCC 13525) and S. epidermidis (ATCC 14990) were obtained from the American Type Culture Collection (ATCC, Rockville, MD, USA). A laboratory culture of E. coli (strain DH 5oc) was used.

P. fluorescens, S. epidermidis, and E. coli have been utilized as challenge microorganisms in many studies reported in the literature. The first two species are common in indoor air environments and relatively easy to handle under laboratory conditions, which helps ensuring accurate and credible experimental data. The selected specie of Pseudomonas and Staphylococcus have relatively low pathogenicity as compared to many other species of the same genus. Generally, there are many microorganisms with limited pathogenicity that have the same (or similar) aerodynamic and/or biochemical characteristics as highly pathogenic agents. Simulants are widely used for evaluating the bactericidal effects. For instance, Bacillus subtilis var niger (BG) spores are well-known simulants of Bacillus anthracis (causing Anthrax) and have been utilized in many studies conducted by the, US Department of Defense and other agencies. The third bacterium selected for the tests, E. coli, is a very sensitive Gram negative bacterium, known to cause various health problems and widely used as a test microorganism in microbial studies [9, 11]. E. coli species used in this study is not pathogenic although it has physiological properties similar to its well-known "cousin" food-borne E. coli O1S7:H7. Some E.coli strains produce enterotoxins. Airborne E. coli have been found in occupational air environments. e.g., in farms [19]. Since a recent study implicated airborne spread of E. coli O1S7:H7 during an outbreak investigation [16] we believe that inclusion of E. coli bacteria as a test microorganism is particularly timely.
Vegetative cells of P. fluorescens and S. epidermidis were cultured by incubating them in Trypticase Soy Broth at 28°C for 18 hours and at 37°C for 24 hours, respectively. Vegetative cells of E. coli were cultured for 18 hours in Luria Bertani (LB) broth at 37°C. After incubation, the vegetative cells were washed three times with sterile deionized water by vortexing (Vortex Touch Mixer, model 231, Fisher Scientific, Pittsburgh, PA, USA) followed by centrifugation at 7,000 rpm for 7 minutes at room temperature (Sorval RC-SB, Sorval Co., Newtown, CT, USA). Before using the bacterial suspension for aerosolization, the concentration of bacteria in the suspension was adjusted to 108 - 109 bacteria/mL, as determined by microscopic counting. After the second washing, the cells were stored at room temperature for up to 3 hours. One more washing was done when refilling the nebulizer with bacterial suspension prior to each subsequent experiment.

The Collison nebulizer aerosolized bacteria-containing water droplets ofup to 3 µm in aerodynamic diameter. Thus, bacterial cells were first encapsulated in water droplets. This simulates the way in which infectious microorganisms often enter the air environment from their sources, such as human saliva and mucus during coughing or sneezing. Then the effluent aerosol was diluted and dried immediately by the temperature- and humidity-controlled sheath air flow. Finally, the dry airborne bacterial cells entered the bioaerosol test chamber at T = 26:!:2°C and RH=l7±5% (dry indoor air). While the bactericidal efficiency was determined primarily under dry air conditions, one experiment with AS150MM and P. fluorescens was conducted at high humidity (RH=89±3%.)
The same bacterial suspension was used for less than 35-40 minutes. This allowed us to minimize the effect of aerosolization time on the initial viability, so the latter remained the same throughout the test.
Once the bioaerosol concentration reached the desirable level inside the chamber and remained at that level for at least 3 min (as measured by the OPC), the collection of airborne bacteria into the BioSampler began. The sampling time was 10 min. Each experiment was performed with three replicates using the same bacterial species with and without running the tested ionic air purifier.
The concentration of viable bacteria in the sample (CFU/mL) was determined by cultivation of the liquid collection medium on nutrient agar plates. Three dilutions (10’1, 102 and 10 -3 were prepared from the original sample. Aliquots of 100 µL from the original suspension and the dilutions were cultivated on agar plates in triplicate. P. fluorescens and S. epidermidis were cultivated on Trypticase Soy Agar, whereas E.coli was cultivated on LB agar (using the following amounts: LB Broth= 10 g; Agar = 7.5 g; Distilled water = 500 mL). The inoculated culture plates were then incubated at 28°C for 40 hours for P. fluorescens, at 37°C for 24 hours for S. epidennidis and at 37°C for 18 hours for
E. coli. The colony forming units (CFU) in each culture plate were counted from the diluted sub-samples that had about 30-300 colonies. The concentration of culturable bacteria in the BioSampler liquid, CCFU (CFU/mL), was calculated as follows:
![]()
where NCFU is the average CFU number determined from three repeats, n is the dilution factor and v0 is the volume of the suspension spread on each agar plate (0.1 mL).
Total (viable plus non-viable) bacterial count was conducted by epifluorescence microscopy (Leitz, Laborlux S, W. Nuhsbaum Inc., McHenry, IL, USA). To determine the total bacterial count in each sample collected by the BioSampler, an aliquot from that sample was filtered and analyzed using acridine orange staining and epifluorescence microscopy.
Prior to filtering the bioaerosol sample suspension through a black polycarbonate filter (Millipore GTBP 0250; pore size 0.2 µm, diameter 25 mm), each filter was equilibrated by filtering 5 mL of sterile phosphate buffer through it Then 5 mL of acridine orange solution (0.1 mg/mL) was added to the bioaerosol sub-sample (taken from the original sample and three dilutions: 10 -1, 10-2 and 10-3) and mixed thoroughly by shaking. The volume of the bioaerosol sub-sample to be enumerated (ranged from 0.2 to 2 mL) was chosen after preliminary tests conducted at various dilution ratios. The volume that resulted in the number of counts ranging from 4 to 40 per microscopic field was selected. After adding the acridine orange stain to the sub-sample the suspension was allowed to stand for 5 minutes and then filtered by vacuum suction. The filter was mounted on a microscopic slide with light mineral oil and a cover slip.
The microorganisms on the filter were counted by the epifluorescence microscope at a magnification of 1000X. For each test, we counted either 40 randomly chosen microscopic fields or a total of 400 bacteria. In the latter case, at least 20 fields were counted. The total bacterial count, CTOTAL, (Number/mL) in the BioSampler suspension was determined as follows:
![]()
where NTOTAL is the average bacterial count per microscopic field, R is the effective radius of the filter (10 mm), A is the area of the microscopic field (0.02351 mm2) and v is the volume (in mL) of the original bacterial suspension analyzed.
The liquid samples were collected from the Collison nebulizer before and after aerosolization. These samples were analyzed for culturable and total counts of bacteria as described above. Five dilutions of the samples (10-3 to l0 -7) were used for these analyses.
After analyzing the culturable and total counts in each sample, we determined the bacterial viability, V, as a ratio of Ccru/CTOTAL The bactericidal effect of the ionic air purifier was quantified as the bacterial inactivation (in percent), calculated by using the bacterial viability data as follows:
![]()
Here VOFF was the bacterial viability fraction obtained when the ionic air purifier was not operating, and VON was the bacterial viability fraction when the ionic air purifier was operating. The average values of the bacterial inactivation and standard deviation from three different experiments were calculated. It is important to stress that our study design accounted for the natural viability loss due to desiccation, thus allowing us to distinguish the loss of viability due to the air ionization.
Results and discussion
Physical efficiency
Figure 4 shows the air ion concentration measured in the large walk-in chamber at a distance of 1 m from the emission point of Minimate, AS180i*. It is seen that the ion concentration increased very rapidly to approximately 3.5x105 ions per cm3 and remained approximately at that level during the entire operation time. Once the ion emitter was turned off, the air ion concentration decreased almost as rapidly as it had increased and reached the initial level. The data suggest that the process of particle charging by ions is homogeneous, which is important for the validation of experimental protocol. The Minimate AS150MM* unit showed similar results, except the "saturation" level was much lower. approximately 2x I04 ions per cm3

Figure 5 presents the time evolution of the nondimensional concentrations of three particle size fractions (smoke particles) in the large walk-in chamber, as measured by the ELPI. The upper curves represent natural decay, and the lower curves represent the decay when the Minimate AS180i/AS150MM ion emitter operated in the chamber. The data obtained with the Grimm OPC for these particle sizes confirmed the ELPI data within ±30%. We found this agreement acceptable given the accuracy of the two instruments and the difference between the aerodynamic and optical sizes of the particles. The data collected with airborne bacteria and NaCl bad the same trends. Quantitatively, the removal rate of bacterial cells followed the one obtained for 1-µm smoke particles (Fig. 5b) with an average deviation of ±18%. The results obtained for smoke and NaCl were as close as ±7%. It is seen that the ion emission results in much more rapid particle removal from the air than the natural air cleaning (due to gravitational sedimentation). For instance, the ion emission decreased the concentration of 0.5 µm particles by a factor of 5 during an hour whereas the respective decrease caused by the natural decay was only 25% (Fig. 5a). The effect of ion emission is rather high, given that it is achieved by a small, battery operated unit (about 10 cm in size) inside an air space as large as 24.3 m3. As the lower curves in Figures 5a-c are almost identical the data suggest that the physical efficiency is about the same for particles in the entire bacterial size range (0.5-2 µm).

The air volume was found to be a factor affecting the physical efficiency of the ionic air purifiers. Figure 6 demonstrates the nondimensional aerosol concentration as a function of the test chamber volume and the time of ion emission. The graphs are based on the particle size integrated data obtained with smoke particles (large walk-in chamber) and NaCl (small walk-in chamber) particles. The aerosol measurements performed with the NaCl using the Aerosizer and Grimm OPC in the small walk-in chamber revealed similar trends. The difference between the data provided by these instruments did not exceed ±20%. Therefore, further tests that involved PSL particles and P. fluorescens bacteria were conducted using the OPC only. The ion emission by AS180i* during the time as short as 3 min does not seem to be sufficient to provide substantial air cleaning even in relatively small air volumes. The data obtained at t = 15 min show about l :5-fold decrease of the initial aerosol concentration of bacterial or bacteria-size particles in the large walk-in chamber (24.3 m3) and 2-fold decrease in the small walk-in chamber (2.6 m3). The ion emission in a very small air space (26 L box) creates tremendous air cleaning effect so that the number of particles that remains airborne in 15 minutes does not exceed few percent of their initial number in that space. The physical efficiencies of AS150MM obtained in the small and large walk-in chambers, respectively, were considerably different at t = 30 and 60 min.
Although the physical efficiency of AS150MM demonstrated the same trends, the decay caused by the ion emission by AS180i* was not as rapid as that obtained with AS180i*, which reflects the difference in their ion emission rate.
Bactericidal efficiency
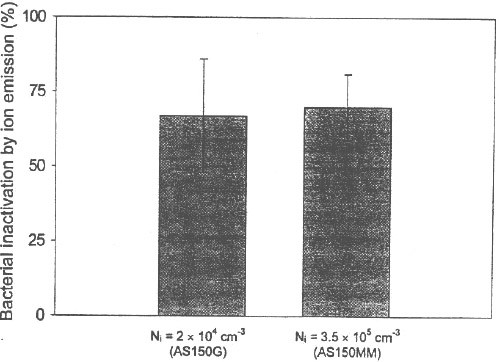
The mean bacterial inactivation values (in percent) and the standard deviations for three species tested in this study at RH=17±5% were: S. epidermidis: 53±20%; P. fluorescens: 71±11% and E. coli: 93±2% (see Fig. 7). The viability of all three microorganisms was affected even by a short exposure to the air ion flow (texp ≈ l min). The bactericidal effect was found to be higher for the two Gram-negative bacteria (P. fluorescens and E. coli) than for the Gram-positive S. epidermidis. The difference between the average inactivation values observed for S. epidermidis and P. fluorescens is relatively small, whereas the average inactivation for E. coli is significantly higher than for the two other species. The data reflect the individual bacterial responses to the stress resulted from the interaction of air ions with bacterial cells and suggest that P. fluorescens and E. coli are more sensitive to injuries caused by electrical charges than S. epidermidis. The lower bacterial inactivation of S. epidermidis cells can be explained by high resistance of Gram-positive bacteria to various types of stresses. The cell wall of Gram-positive bacteria is rigid [13] and thick, thus protecting the bacterial cells from environmental stresses. In contrast, Gram negative bacteria of P. fluorescens and E. coli have very thin sheet-like cell envelopes [12] that offer less protection against environmental stresses. The difference in bacterial responses to unipolar electric charges found between the Gram-negative and Gram-positive cells might also have been caused by the chemical differences in their cell wall structure. The cell wall of Gram-negative bacteria has more lipid content, whereas Gram-positive bacteria have more peptidoglycans in the cell wall. This may differently affect the charge-related orientation of metabolically linked proteins and other cell membrane components. Figure 8 presents the comparative bactericidal effect of the Minimate AS180i/AS150MM* ionic air purifiers obtained with P. fluorescens cells at RH = 17±5% (texp ≈ 1 min). With the bacterial inactivation of 69±20% for ASlSOG and 71±11% for AS150MM, no stastically significant difference (t-test: p>0.05) was observed for these ion emitters, although they produce significantly different ion concentration levels. The data suggest that while the difference in the ion emission rate did affect the physical (particle removal) efficiency, it appeared to have: no effect on the bactericidal efficiency under our experimental conditions.

The bactericidal effect caused by the air ions on viable airborne cells of P. fluorescens after the I-min exposure decreased when the humidity level increased from 17±5% to 89±3%. The average value of the bacterial inactivation dropped more than 3-fold and the data variability increased considerably.

Thus, the bactericidal effect of the air ion emission is more pronounced at low air humidity, typical for indoor environments. Since the bacteria may become coated by a thin layer of water when exposed to a high humidity environment, we hypothesize that this layer could shield the bacterial cell wall from the air ions.
Combined effect
The data on the physical and bactericidal efficiencies of the air ion emission suggest that the reduction in the indoor aerosol concentration combined with the bacterial inactivation can sjgnificantly reduce the human exposure to indoor air pollutants, such as.particles and microorganisms.. The.following estimate was made based on the data obtained with AS150MM* operated during 30 minutes in. the small walk-in chamber: as about 80% of viable airborne bacteria have been removed from indoor air (Fig. 6) and.at least 71% of the cells remaining in the air have lost their viability during the same time (P. fluorescens, t > l min, Fig. 7), the overall efficiency of the ion emission against the viable bacterial agent is 1- (1 - 0.8)(1 - 0.71) = 0.942, i.e. 94.2%. This corresponds to an almost 20-fold exposure reduction.
Disclaimer
Reference to any companies or specific commercial products does not necessarily constitute or imply their endorsement, recommendation, or favoring by the group of authors or by the University of Cincinnati.
*This document originally pertained to the AS150G and AS150MM, Wein Products, Inc
