Indoor Air 2005
www.blackwellpublishing.com
S. A. Grinshpun, G. Mainelis, M. Trunov, A. Adhikari, T. Reponen, K. Willeke
Center for Health-Related Aerosol Studies, Department of Environmental Health; University of Cincinnati, Ohio, USA. Present address Department of Environmental Sciences Rutgers University, New Brunswick, New Jersey, USA. Present address Mechanical Engineering Department, New Jersey, USA. Institute of Technology, Newark, New Jersey. USA.
Abstract
Numerous techniques have been developed over the years for reducing aerosol exposure in indoor air environments. Among indoor air purifiers of different types, ionic emitters have gained increasing attention and are presently used for removing dust particles, aeroallergens and airborne microorganisms from indoor air. In this study, five ionic air purifiers (two wearable and three stationary) that produce unipolar air ions were evaluated with respect to their ability to reduce aerosol exposure in confined indoor spaces. The concentration decay of respirable particles of different properties was monitored in real time inside the breathing zone of a human manikin, which was placed in a relatively small (2.6 m³) walk-in chamber during the operation of an ionic air purifier in calm air and under mixing air condition. The particle removal efficiency as a function of particle size was determined using the data collected with a size-selective optical particle counter. The removal efficiency of the more powerful of the two wearable ionic purifiers reached about 50% after 15 min and almost 100% after 1.5 h of continuous operation in the chamber under calm air conditions. In the absence of external ventilation, air mixing, especially vigorous one (900 CFM), enhanced the air cleaning effect. Similar results were obtained when the manikin was placed inside a partial enclosure that simulated an aircraft seating configuration. All three stationary ionic air purifiers tested in this study were found capable of reducing the aerosol concentration in a confined indoor space. The most powerful stationary unit demonstrated an extremely high particle removal efficiency that increased sharply to almost 90% within 5-6 min, reaching about 100% within 10-12 min for all particle sizes (0.3-3 µm) tested in the chamber. For the units of the same emission rate, the data suggest that the ion polarity per se (negative vs. positive) does not affect the performance but the ion emission rate does. The effects of particle size (within the tested range) and properties (NaCl, PSL, Pseudomonas fluorescens bacteria) as well as the effects of the manikin's body temperature and its breathing on the ionic purifier performance were either small or insignificant. The data suggest that the unipolar ionic air purifiers are particularly efficient in reducing aerosol exposure in the breathing zone when used inside confined spaces with a relatively high surface-to-volume ratio.
Practical Implications
Ionic air purifiers have become increasingly popular for removing dust particles, aeroallergens and airborne micro organisms from indoor air in various settings. While the indoor air cleaning effect, resulting from unipolar and bipolar ion emission, has been tested by several investigators, there are still controversial claims (favorable and unfavorable) about the performance of commercially available ionic air purifiers. Among the five tested ionic air purifiers (two wearable and three stationary) producing unipolar air ions, the units with a higher ion emission rate provided higher particle removal efficiency. The ion polarity (negative vs. positive), the particle size (0.3-3 µm) and properties (NaCl, PSL, Pseudomonas fluorescens bacteria), as well as the body temperature and breathing did not considerably affect the ionization-driven particle removal. The data suggest that the unipolar ionic air purifiers are particularly efficient in reducing aerosol exposure in the breathing zone when they are used inside confined spaces with a relatively high surface-to-volume ratio (such as automobile cabins, aircraft seating areas, bathrooms, cellular offices, small residential rooms and animal confinements). Based on our experiments, we proposed that purifiers with a very high ion emission rate be operated in an intermittent mode if used indoors for extended time periods. As the particles migrate to and deposit on indoor surfaces during the operation of ionic air purifiers, some excessive surface contamination may occur, which introduces the need of periodic cleaning these surfaces.
Introduction
Inhaled airborne particles and microorganisms can cause adverse health effects, such as asthma and allergic diseases (Burge, 1990; Koskinen et al., 1995; Miller, 1992; Spengler et al., 1993) as well as airborne infections (Burge, 1990). Exposure to indoor aerosol pollutants has become a growing public and occupational health concern (American Lung Association, 1997; Gammage and Berven, 1996; Samet and Spengler, 1991). The outbreaks of emerging diseases and the threat of bioterrorism have generated special needs in indoor air cleaning against respirable particles, especially those of biological origin. Strategies developed for protecting building environments from deliberately used aerosol agents require efficient air filtration and air cleaning systems [National Institute for Occupational Safety and Health (NIOSH), 2003]. Conventional indoor air purifiers include mechanical filters, electronic air cleaners, hybrid filters, gas phase filters and ozone generators. Among various mechanisms, the emission of ions, also referred to as air ionization, has shown considerable promise. Emission of bipolar ions enhances the agglomeration of smaller particles into larger ones, which then gravitationally settle and thereby purify the air. Ionization may also cause attraction between particles and grounded surfaces resulting in electrostatic deposition.
The physical and biological effects of small air ions on indoor air quality as well as various health benefits and implications of air ionization have been discussed in the literature (Daniell et al., 1991; Kondrashova et al., 2000; Krueger and Reed, 1976; Soyka and Edmonds, 1977; Van Veldhuizen, 2000; Wehner, 1987). The ion emitters, which meet health standards (e.g. by not generating ozone above the established thresholds), have been incorporated in commercial air purification devices that utilize either bipolar or unipolar ion emission. These devices are currently produced by several manufactures worldwide (Sharper Image Inc., Little Rock, AR, USA; Topway Electronic Factory Co., Guangzhou, China; Wein Products, Inc., Los Angeles, CA, USA; etc.) and used in residential and occupational settings for removing dust particles, aeroallergens, and airborne microorganisms from the air. The ion emission has been tested by several investigators for its ability to reduce the indoor aerosol concentration (Bigu, 1983; Bohgard and Eklund, 1998; Grabarczyk, 2001; Harrison, 1996; Hopke et al., 1993; Khan et al., 2000; Kisieliev, 1966; Li and Hopke, 1991). The bactericidal effect of air ionization has also been assessed (Lee, 2001; Marin et al., 1989; Seo et al., 2001; Shargavi et al., 1999). However, the mechanisms involved in the ionic purification of inhaled air in the breathing zone remains poorly understood. Furthermore, there are still controversial claims (favorable and unfavorable) about the performance of commercially available ionic air purifiers. This controversy reflects a lack of quantitative data on the purifiers’ efficiency in peer-reviewed scientific journals.
Ionic air purifiers are available as stationary and wearable devices. The latter have been specifically developed for personal respiratory protection by targeting particles in the human breathing zone. Some models are designed to operate in confined spaces, such as automobiles, aircraft cabins, bathrooms, office cubicles, and small animal confinements. Our pilot study has demonstrated that unipolar ion emission by corona discharge may considerably reduce the aerosol concentration in the breathing zone (Grinshpun et al., 2001). We concluded that the concentration decrease, during the air ionization, occurs as air ions impart electrical charges of the same polarity on aerosol particles, and the unipolarly-charged particles then repel each other out of the breathing zone towards nearby surfaces, where they are deposited. More recent investigation by our group (Lee et al., 2004) has shown that a high-density emission of unipolar ions has a good potential for air cleaning across room-size indoor spaces uniformly contaminated with fine and ultrafine aerosol particles. Another recent work - an extensive theoretical study of Mayya et al. (2004), which was awaiting publication when the present paper was being completed - has identified and analyzed several physical factors affecting the airborne particle removal by unipolar ionization and developed advanced computational model to quantify the process.
In this study, we determined the particle removal efficiencies of five ionic air purifying devices - two wearable and three stationary units - that produce unipolar ions (either positive or negative). The concentration decay of respirable particles (0.3- 3 μm) was monitored in real time inside the breathing zone of a human manikin placed in a chamber that simulated a confined indoor environment. The role of air mixing in the chamber as well as the breathing effect and the body temperature effect on the performance of the ionic air purifiers were also investigated.
Study design and methods
Test room
The tests were conducted in a walk-in chamber made of wood (painted) with interior dimensions of L x W x H = 1.2 m x 1.0 m x 2.2 m ≈ 2.6 m³. A standard size human manikin was placed inside facing the chamber center, see Figure 1. The manikin’s nose was located 0.3 m from the back of the chamber, 0.5 m from the side walls, and 1m from the floor (sitting position). This configuration was used in our previous studies on evaluations of respirators (Willeke et al., 1996).
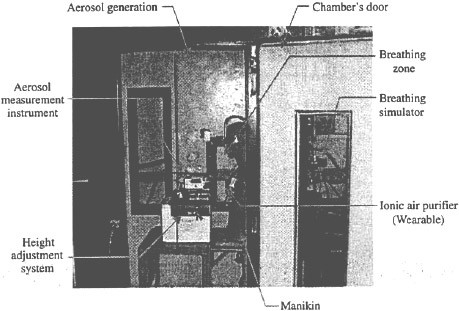
Aerosol generation and flow patterns
Polydisperse NaCl particles and monodisperse poly styrene latex (PSL) spheres, as well as bacterial cells of Pseudomonas fluorescens were used as test aerosols. Both types of biologically inert particles (NaCl and PSL) and P. fluorescens bacteria have been extensively utilized in earlier studies for evaluating sampling devices and respirators (Grinshpun et al., 1999; Mainelis et al., 2002a,b; Stewart et al., 1995; Terzieva et al., 1996; Wang et al., 2001; Willeke et al., 1996). Gram-negative P. fluorescens bacteria are commonly found in air environments. A standard Collison nebulizer (BGI Inc., Waltham, MA, USA) operated at a flowrate of 6 1/min was employed to aerosolize the test particles from a liquid suspension. The liquid content of the aerosolized droplets was evaporated by mixing the aerosol flow with 80 1/min of dry filtered air. The combined airflow entered the test chamber through an air laminarizing and distributing unit. The air exhaust, positioned at the bottom of the chamber, was connected to an external pump through a HEPA filter.
Most of the tests were performed with NaCl aerosol generated from a solution prepared by dissolving 20 g of reagent quality NaCl (Fisher Chemical, Fair Lawn, NJ, USA) into 400 ml of deionized and sterilized water. After drying, the particles had a broad size spectrum, including the range of interest [0.3- 3 μm as measured by an optical particle counter (OPC)]. This range represents a wide variety of aeroallergens and microbial agents (Reponen et al., 2001).
Two size fractions of PSL spheres (Bangs Laboratories, Inc., Fishers, IN, USA), with median count diameters of 0.44 µm (ag = 1.07) and 0.95 µm (ag = 1.1), were used for selected experiments. Prior to aerosolization, the suspension of PSL particles was deagglomerated for 5 min in an ultrasonic bath (model 220, Branson Cleaning Equipment Co., Shelton, CT, USA). Bioaerosols of P. fluorescens (ATCC 13525; American Type Culture Collection, Rockville, MD, USA), rod-shaped bacterial cells of dₚ ≈ 0.8 µm, were also used for selected experiments. Standard microbial preparation procedures utilized in our previous studies (Mainelis et al., 2002a,b; Stewart et al., 1995; Wang et al., 2001) were followed prior to aerosolizing the P. fluorescens cells. The tests with PSL and P. fluorescens were performed to study the effect of the physical material and biological status of particles on the particle removal by continuous air ion emission.
Aerosol monitoring
During each test, the concentration and size distribution of the airborne particles were monitored in real time with an OPC (model 1.108; Grimm Technologies Inc., Douglasville, GA, USA). It enumerated aerosol particles every minute in 16 detection channels within the particle size range of 0.3 to about 30 µm. The range of interest was represented by the following eight channels (the size fractions are listed by their channel mid-points): < dp > = 0.35, 0.45, 0.58, 0.73, 0.90, 1.3, 1.8 and 2.5 µm.
A wearable air purifier was positioned on the manikin’s chest so that the ion emission point would be in line with the manikin’s nose, which was 0.2 m above the purifier. The OPC inlet was positioned directly above the purifier’s outlet, perpendicular to the line between the purifier and the manikin’s nose. The aspiration efficiency of the OPC inlet is approximately l00% regardless of its orientation since the test particles (0.3-3.0 µm) are virtually inertialess (Grinshpun et al., 1990, 1993). At first the OPC measurements were performed at three locations: 1, 10 and 19 cm above the ion emission point. Although the variability of the aerosol concentration near the ionizer was very high, the aerosol concentrations measured at 10 cm (upper chest level) and 19 cm (nose-mouth area) were essentially the same with a variability ranging from 10 to 20% for the entire tested particle size range. We concluded that it would be sufficient to measure the aerosol at the point representing inhaled air.
When evaluating the performance of stationary air purifiers, aerosol sampling was also performed in the breathing zone, while the tested unit was placed either on the floor or on the table (in accordance with its operational function). Overall, the inhalation point seems the most appropriate location for the OPC inlet as the ultimate objective is to characterize the air purification effect of ionic emitters in terms of respiratory exposure to airborne contaminants. Our preliminary study revealed no significant effect of the distance from the emission point to the walls of the test chamber on the aerosol concentration as long as this distance exceeded 0.45 m. Stationary air purifiers were evaluated at equal distance between the manikin’s face and the opposite wall.
Determination of the particle removal efficiency
The size-specific aerosol concentration, C (dp), was measured as a function of monitoring time, t. The natural decay was measured for 10 h as a baseline test. In the experiments involving air ion emission, the time varied from 1 to 3 h, depending on the emission rate of the ionic air purifier being tested. For each purifier, two concentration decay curves were obtained: the natural decay, i.e. when the ion emitter was ‘off’ [Cnatural (dp, t)], and the one with the ion emitter ‘on’ [Cionizer (dp, t)]. The particle removal efficiency was determined as follows:

The natural decay depends on the air mixing conditions in the chamber. Therefore, separate baseline tests were conducted in calm air as well as with the fan operating at 33 and 900 CFM.
It should be noted that the above definition is different from the ratio of the initial to the final aerosol concentration levels, which is referred to as the ‘concentration reduction factor (CRF)’ and often used in the literature. Mayya et al. (2004) acknowledged the limitation of CRF indicating that it ‘is not a primary index of ionizer performance.’ The particle removal efficiency used in this study allows comparing the ionization-driven concentration decrease to the natural decay occurring because of gravitational settling, diffusion and other mechanisms.
Experimental procedure
Prior to each experiment, the test chamber was ventilated by supplying particle- and ion-free air for about 1 h, until the total particle concentration inside the chamber was below 10³ particles per liter of air. At that time, the ventilation was turned off and the aerosol generation system was activated. The fans operated at two points inside the chamber to achieve uniform aerosol concentration across the volume. Once the total concentration reached about 10⁶ particles per liter of air, the aerosol generator and the fan were turned off. After waiting for 5 min to allow the concentration to stabilize, the test began (t = 0). When using a non-breathing manikin under calm air-conditions, the only device that was operating inside the chamber at t > 0 was the OPC, which ran at a very low flow rate (1.2 1/min) so that no considerable air movement occurred. When testing with a breathing manikin or under air mixing conditions, the breathing simulation machine or/and the air mixing fan produced significant air movement inside the chamber. The tests involving the breathing manikin were conducted to determine whether the inhalation-exhalation cycle affects the particle removal efficiency of the tested ionic air purifier. The air flow was supplied by a breathing simulation machine located outside the test chamber. The aerosol concentration decay solely resulting from the breathing simulation was compared with the natural aerosol decay in the chamber. The tests were also conducted with the manikin pre-heated to an average temperature of T = 40°C. These experiments were carried out for 1 h to assess whether the body temperature affects the performance of the ionic purifier. The aerosol concentration decay measured with the preheated manikin was compared with the results obtained with a non-heated manikin (T = 23°C). The manikin was made of non-conductive material and dressed in a laboratory coat (not shown in Figure 1), which was washed between the tests.
A separate set of experiments was performed with the manikin placed in a partial enclosure built inside the test chamber. This confined space was restricted by front, side and overhead panels. The configuration simulated a passenger seating section in an aircraft (front panel = front seat, side panels = nearby passengers, overhead panel = overhead compartment). The volume of the partially enclosed air space was about 0.275 m³ consisting of about 0.250 m³ of open space in the front and on the sides of the manikin and about 0.025 m³ of open space between the manikin’s head and the overhead panel. Gaps of 7.5 cm between the panels allowed air exchange. To evaluate the effect of indoor air volume on the particle removal efficiency, one wearable unit placed in a very small Styrofoam chamber-box (L x W x H = 0.31 m x 0.30 m x 0.28 m ≈ 0.026 m³ = 1/100 of the main test chamber). The purifier was located at the midpoint of the bottom surface (the small dimensions of the chamber-box did not allow using the manikin inside the box). The OPC inlet was positioned 10 cm above the ion-emission point.
Tested air purifiers
Two models of wearable ionic air purifiers, provided by Wein Products, Inc. (Los Angeles, CA, USA), were tested in this study. One was equipped with a metal grid acting as an electrostatic precipitator, the Minimate prototype* (further referred to as W1), and the other had no grid, the Minimate prototype*(further referred to as W2). Both units emitted positive ions with the production rate of W2 being five- to l0-fold higher than that of W1. The estimated ion densities produced by these devices (based on the measurement data reported by the manufacturer) were in the range of ~0.5 x l0⁵ to ~5 x 10⁵ ions per cm³ at a distance of 1 m from the emission point, assuming calm air conditions. Three stationary units (also by Wein Products, Inc.) were the AS250B* (further referred to as S1) with an estimated ion density of ~2 x 10⁵ positive ions/cm³ at 1 m from the emission point, the AS1250B* (referred to as S2) with ~5 x l0⁵ positive ions/cm³ and the VI-3500 (referred to as S3) with ~30 x l0⁵ negative ions/cm³. For each set of conditions, three replicate tests were performed. Although the variability of the measured aerosol concentration was almost 20% for NaCl and PSL particles and about 25% for bacteria, the removal efficiency showed very low variability (usually < 5%). The data were statistically analyzed by using the Microsoft Excel software package (Microsoft Co., Redmond, WA, USA).
Results and discussion
Natural decay of airborne particle concentration
In the absence of ion emission, the aerosol concentration in the test chamber slowly decreased with time.The natural air cleaning mechanisms in calm air contaminated with fine particles (about 0.1- 2 µm) are primarily gravitational sedimentation and diffusion. For the tested particle size range of 0.3- 3 µm, the natural aerosol concentration decay is driven mainly by sedimentation. The measurement data showed that the concentration of 2- 3 µm NaCl particles in the test chamber naturally decreased by 50% in about 2 h, while for 1 µm particles of NaCl and P. fluorescens cells, the 50% decrease took over 10 h. The concentration of 0.3-0.4 µm particles of NaCl showed < 10% decrease during a l0-h period. Most of our experiments involving ionic air purifiers were conducted within much shorter time periods (1-3 h), because our pilot data showed that the air ionization can significantly reduce the aerosol concentration during about an hour (Grinshpun et al., 2001).
Wearable ionic air purifiers
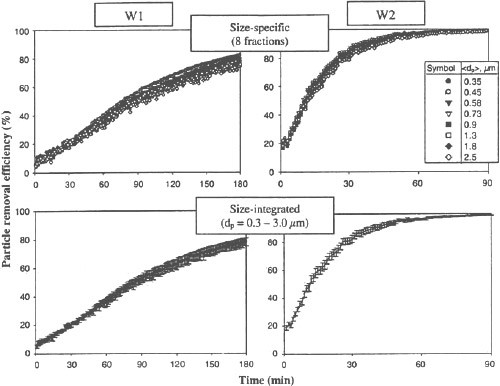
Figure 2 shows the particle removal efficiency as a function of time for the W1 and W2 wearable ionic air purifiers operating in calm air. The top two figures show the size-selective measurement data obtained with NaCl particles and recorded in the first eight OPC channels. The bottom figures show the size-integrated data for the entire measured particle size range of 0.3-3 μm. The data suggest that the removal efficiency did not have a clear trend with the particle size (within the tested range). The particle removal efficiency of W1 increased gradually from 5-15% at t = 15 min to about 30-40% at t = 1 h. The size-integrated data demonstrate that the air cleaning provided by this purifier reached considerable levels after it had continuously operated in the chamber for more than an hour: the aerosol concentration of NaCl in the breathing zone decreased by a factor of 2 (removal efficiency = 50%) after 1.5 h and almost fivefold (removal efficiency = 80%) after 3 h. The particle size-specific removal efficiency values lay within a 15% corridor from each other at each specific time point.
The purifier labeled as W2 provided much more efficient air cleaning as compared with W1, which can be attributed to a higher ion production rate. The particle removal efficiency of W2 reached approximately 50% during the first 15 min and continued increasing with the time. As a result of its 1.5-h continuous operation on the manikin’s chest, almost 100% of the initially airborne particles were eliminated from the breathing zone. Comparison of the performance characteristics of W1 and W2 under calm air conditions confirms that the air cleaning provided by a unipolar ion emission becomes more efficient at a higher ion production rate.
When the fan was operated in the test chamber at 33 CFM, it produced an air velocity level equivalent to the air exchange rate of about 20 AEH (no external ventilation was actually introduced during the tests). At this air mixing rate, the particle removal efficiencies of both wearable ionic air purifiers were slightly higher than under calm air conditions. However, the difference was not statistically significant (t-test: P > 0.05). Once vigorous air mixing (900 CFM) was introduced, the air cleaning became much more rapid. The air currents intensify the ion propagation in the chamber and enhance the ion-particle interactions. This makes the
particle charging by air ions more efficient and consequently increases the removal efficiency.
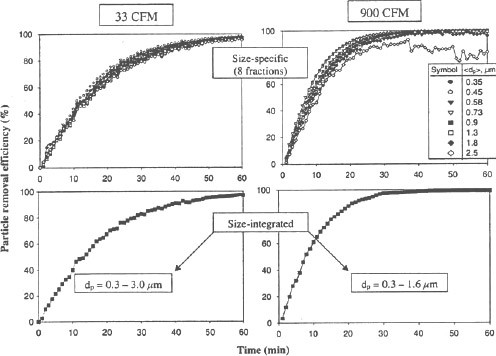
Figure 3 presents the data obtained with W2 operating under air mixing conditions of 33 and 900 CFM. It is seen that during the first 15 min about 50-60% of the airborne particles were removed at 33 CFM and about 65-80% at 900 CFM. The ion emission appeared to be sufficient to clean the chamber air of essentially all the test particles in about 1 h of operation when it was enhanced by a 33 CFM fan in the chamber: the size-integrated aerosol concentration determined for dp = 0.3- 3.0 µm decreased to almost 1% of its initial level. At 900 CFM, all particle size fractions, except the two largest ones, dp = 1.6-2.0 µm (< dp > = 1.8 µm), and 2.0-3.0 µm (< dp > = 2.5 µm), showed the aerosol concentration reduction by a factor of ~10² –10³ in about 40 min. The curves representing < dp > = 1.8 and 2.5 µm show excessive variability because intensive air mixing removed the larger particles from the air very quickly making the baseline concentration measurement statistically unreliable in about 30-40 min. Consequently, the size integrated efficiency at 900 CFM is shown only for dp = 0.3-1.6 µm.
Compared with a typical air exchange rate in an indoor environment (Abt et al., 2000), the air mixing rate of 900 CFM creates rather excessive air movement (especially when it is applied to confined spaces), while 33 CFM seems more reasonable. As the data collected with both wearable purifiers show that the increase from 0 to 33 CFM did not significantly affect their performance, further experiments were performed at 33 CFM. Thus, on one hand, by introducing some air mixing we made our experimental conditions more representative of a typical small room or cabin, and, on the other hand, we kept the air mixing rate below the level at which it begins to significantly affect the particle removal efficiency.
Stationary ionic air purifiers

The particle removal efficiency curves for the three stationary ionic air purifiers are shown in Figure 4. The curves represent the size-selective monitoring data obtained with NaCl particles under air mixing conditions (33 CFM). Each of the three devices demonstrated considerable air cleaning efficiency. The particle removal efficiency of SI shows some dependence on particle size, although this effect was not very pronounced. The size-specific curves are similar to those obtained for the wearable W2 unit, which reflects the fact that their ion production rates are approximately the same. Surprisingly, S2 that has a higher ion production rate than S1 does not show greater particle removal efficiency.
The third stationary purifier, S3, demonstrates extremely high particle removal efficiency: it increased sharply to almost 90% within 5-6 min, reaching about 100% within 10-12 min for all tested particle sizes. This is attributed to its very high ion production rate. When this air ionizer was operating in the test chamber for a prolonged period of time, such as 1 h, the high number of ions emitted in the relatively small volume of 2.6 m³ produced considerable electric fields that resulted in charging of objects inside the chamber, subsequently causing electrostatic discharges. Our observations suggest that these occasional undesirable electrostatic discharges during human activity represent a limitation for the long-term continuous use of powerful ionic devices in confined spaces. The related safety issue may be resolved by introducing a time limit for their continuous operation. Indeed, as the particle removal efficiency of S3 reaches a plateau of about 100% in 10-12 min, there is no need to keep this purifier operating continuously for an hour in an air volume as small as 2.6 m³. We examined the performance of S3 operating in an ‘on-and-off’ alternating mode for a period of 1 h, during which ions were periodically emitted for 10-min with a subsequent 10-min interruption. It was found that the overall particle removal efficiency exceeded 98% after an hour of its operation in this intermittent mode. Although the purifier’s performance remained very high, the electrostatic discharge problem was reduced. We believe that the above strategy can be used in the field if the device is equipped with a timer allowing it to emit air ions in an intermittent mode.
Effect of properties of the test aerosol particles
For all five ionic air purifiers evaluated in this study, the tests performed with PSL spheres and P. fluorescens bacteria confirmed the findings obtained with the NaCl aerosol. When testing with PSL particles (dp = 0.44 and 0.95 µm), the removal efficiency was approximately the same as that of NaCl particles of the same sizes and operational time. However, some difference was observed when testing with airborne bacteria. For instance, the data generated with P. fluorescens cells using W1r were about 15% lower (on average) than those obtained with NaCl and PSL particles and had considerable variability.
Pseudomonas fluorescens cells are rod-shaped and thus can be charged by air ions differently than the spherical PSL particles or close to spherical salt particles. Furthermore, the initial particle charge levels of P. fluorescens cells are likely to be different from the initial charge levels of the nonbiological test particles used in this study. Gittens and James (1963) and Sherbet and Lakshmi (1973) have shown that Gram-negative water-borne bacteria have an overall net negative surface charge because of the presence of ionizable amino (NH₂) and carboxyl (COOH) groups of proteins exposed at the cell surface. The data reported in our earlier publications (Mainelis et al., 2001, 2002c) suggest that the aerosolized P. fluorescens bacteria can carry significant electrical charges (up to 10⁴ elementary charges per individual cell), which sharply contrasts with the low electrical charges carried by airborne NaCl particles.
Effect of microenvironment

In addition to the above-described experiments conducted in our 2.6 m³ walk-in test chamber, the air purifiers were also tested on a manikin placed inside the partial enclosure (0.275 m³) that was placed inside the walk-in chamber to simulate aircraft seating. Figure 5 compares the performances of W1 and W2 in the walk-in chamber (with no enclosure built inside) to those inside the enclosure. The particle removal efficiencies shown here are the size-integrated data obtained with the NaCl aerosol during a 90-min experiment at 33 CFM. The reduction in aerosol concentration was found to proceed at a similar pace with and without the partial enclosure. The particle removal efficiency was somewhat higher inside the enclosure for the W2 unit, although the difference between corresponding size-integrated values for the two typed of enclosure was mostly below 10%. The particle removal efficiency of W2 reached about 40% after 10 min of continuous operation, and about 70%after 20 min. After 40 min it exceeded 90%, and was in the range of 95- 98% between 60 and 90 min. For W1, the particle removal efficiency measured on the manikin inside the partial enclosure was lower than that of the more powerful W2, similar to the results that were obtained in a walk-in chamber with no enclosure.
The performance of an ionic air purifier in a specific microenvironment is believed to depend on the volume of this microenvironment. Given that the air volume inside the partial enclosure is about an order of magnitude lower than that of the full-size walk-in chamber, it was somewhat surprising to find such a small difference between the data obtained in these two settings. At the same time, it can be explained by the fact that the partial enclosure has sizable gaps allowing the ‘enclosed’ air to exchange with the air in the chamber (representing passenger aircraft seating, which is also not fully separated from the air in the cabin). An additional experiment conducted in a very small chamber-box (0.026 m³ revealed that the particle concentration in that box decreased much more rapidly than in the 100-times larger walk-in chamber. For example, when W1 was operated inside this box, the particle removal efficiency reached 50% in about 2 min and 90% in about 8 min. The data collected in the two fully-enclosed microenvironments (0.026 and m³) confirm that the time needed to remove a certain percentage of particles from a fully enclosed microenvironment by unipolar ion emission is greater for the larger air volume. The ion emitter operating in a specific air volume creates a charged aerosol cloud, in which the particles (i) repel each other towards the chamber walls where they subsequently deposit and (ii) create ‘image charges’ (same magnitude, but opposite polarity) in the surrounding non-conductive surfaces so that they are attracted to these surfaces and deposited on them. The wall surfaces of the small box (made of dielectric Styrofoam) promote higher electrostatic deposition of the particles charged by air ions than the painted wooden walls of the walk-in chamber. In addition, the top of the small chamber, which was close to the ion emitter, deflected the ion wind of ~ 0.5 m/s. Resulting from this deflection, the ions were quickly and uniformly distributed throughout the small chamber air volume, causing considerable particle deposition on the wall surfaces.
Effects of breathing and body temperature
One would expect that when a breathing simulator is used with the manikin, the air in the chamber would slowly be cleaned because of the inhalation-exhalation process itself (some inhaled particles will be lost inside the machine). At breathing rate of 30 1/min (medium work load), the entire air volume of 2.6 m³ would circulate through the machine during about 1.5 h, assuming that no air molecule will move in and out twice. At 85 1/min (heavy work load), this would occur during about half an hour. The natural aerosol concentration decays in the breathing zone, measured respectively with and without the breathing machine operating with the manikin, allowed determining the relative contribution of the breathing effect to the particle concentration reduction as a function of time. At 30 1/min, this contribution gradually increased from 0 to about 20-40% in 1 h (with the time-integrated contribution of approximately 15%). At 85 1/min, it reached about 40-60% in an hour (time-integrated contribution 25%). Thus, the aerosol concentration reduction occurring because of the manikin breathing is smaller than the air cleaning effect provided by ion emission. It was concluded that the operation of the breathing machine attached to the manikin has little effect on manikin-based performance evaluation of ionic air purifiers.
To explore the potential effect of body temperature on the ion and particle movement and thus on the particle removal from the breathing zone, we compared the data obtained with non-heated and pre-heated manikins. Pre-heating was chosen over continuous electrical heating since the electric heating system affected the behavior of the airborne ions near the manikin during the test and, thus, the air purifier’s performance. The particle removal efficiencies of the W2 purifier plotted as a function of time were compared for a non-heated manikin maintained at 23°C and a manikin that was pre-heated to maintain its average body temperature at about 40°C for an hour. The size-specific data show that the body temperature has no significant effect on the performance of the ionic air purifier, regardless of particle size (t-test: P > 0.05).
Conclusions and future work
All five tested unipolar ionic air purifiers were shown capable to significantly reduce the aerosol concentration in the breathing zone of a human manikin, especially in a confined space. Air mixing, especially vigorous one (900 CFM), established in a non-ventilated chamber enhanced the air cleaning effect. While observing most efficient air cleaning with the most powerful unipolar ion emitter, we found that its long term (~1 h) operation in a confined space can excessively charge objects in the vicinity causing occasional electrostatic discharges. As the latter may limit long term continuous use of ionic devices in low-volume environments, we propose that purifiers with a very high ion production rate should be operated in an ‘intermittent mode.’ Our test confirmed the feasibility of this approach. Additionally, the use of ionic emitters in an intermittent mode may reduce the ozone level that may be of concern when a powerful ionizer operates during a prolonged period of time (Niu et al., 2001). Another (rather obvious) limitation comes from the fact that the aerosol particles, which migrate to indoor surfaces and eventually deposit there, contaminate these surfaces (extreme cases are referred to as ‘the black wall effect’). Thus, from the practical stand point, the air purification based on the continuous air ion emission adds the need of cleaning of the surfaces periodically to avoid an excessive particle accumulation. In this light, it seems important to investigate the effect of wall materials on the ionization-based aerosol particle removal as well as on the subsequent cleaning of indoor surfaces.
The data suggest that the ion polarity does not affect the performance, but the ion emission rate does. The effects of particle size (within the tested range) and properties (NaCl, PSL, P. fluorescens bacteria) as well as the effects of the manikin’s body temperature and breathing on the ionic air purifier performance were either small or insignificant. It seems that the role of particle size should be further investigated. Experimental study of Grabarczyk (2001) and theoretical work of Mayya et al. (2004) indicate that the CRF of an ionic air purifier is particle size dependent. On the other hand, in contrast to CRF, the particle removal efficiency defined by Equation 1 may have shown a suppressed size effect because both of its components, [Cionizer (dp, t)] and [Cnatural (dp, t)], are particle size dependent.
The unipolar ionic air purifiers seem to be more efficient in reducing aerosol exposure in the breathing zone when used in confined spaces characterized by a small volume and -- as a result -- by a relatively high surface-to-volume ratio (such as automobile cabins, aircraft seating areas, bathrooms, cellular offices, small residential rooms, and animal confinements). More data are needed to better predict the performance of ionic purifiers in rooms with different surface-tovolume ratios.
Another issue that deserves further investigation is a situation when continuous particle generation occurs in indoor air environment during ion emission. Our tests presented in this paper were conducted when the aerosol concentration was not sustained by a particle source; therefore, the concentration depleted essentially to zero after certain time. However, if the particle source is present, the concentration should attain a steady-state level, which is generally different from zero (Mayya et al., 2004). This level can be determined from the balance equation accounting for the velocity of the particle migration to indoor surfaces and the particle production rate by the source. In this work, we did not “intend” to study the effect of initial aerosol concentration on the performance of ionic air purifiers. Thus, the initial concentration was not a subject of considerable variation in our tests. However, theoretical modeling conducted by Mayya et al. (2004) for the background levels covering a six order of magnitude range has revealed that the particle charging process depends on the initial aerosol concentration. As a result, at high initial concentration, the concentration reduction should occur rapidly in the beginning (because of a considerable space charge effect) and then attain near linearity as the concentration further decreases. At relatively low initial concentrations, a constant removal rate should be expected resulting in approximately linear reduction in a log-linear scale (Mayya et al., 2004). More experiments are needed to address this issue.
Acknowledgement
The authors are grateful for the cooperation of Wein Products, Inc., which helped initiate this research and made available various air purifying devices to the project. The technical assistance provided by Mr. Dainius Martuzevicius during the manuscript preparation is also appreciated.
*The tested units are prototypes of the currently available Minimate Personal Air Purifier AS150MM that has an ion production rate of 1.2 x 10¹⁴ ions per second as reported by the manufacturer.
* Sanimate Washroom Ionic Purifier AS250B
*Automate Car Plug-in Ionic Purifier AS1250
*Vortex Desktop Ionizer VI-2500
